Lead acid batteries were invented in 1859 by Gaston Planté and first demonstrated to the French Academy of Sciences in 1860. They remain the technology of choice for automotive SLI (Starting, Lighting and Ignition) applications because they are robust, tolerant to abuse, tried and tested and because of their low cost. For higher power applications with intermittent loads however, Lead acid batteries are generally too big and heavy and they suffer from a shorter cycle life and typical usable power down to only 50% Depth of Discharge (DOD). Despite these shortcomings Lead acid batteries are still being specified for PowerNet applications (36 Volts 2 kWh capacity) because of the cost, but this is probably the limit of their applicability and NiMH and Li-Ion batteries are making inroads into this market. For higher voltages and cyclic loads other technologies are being explored.
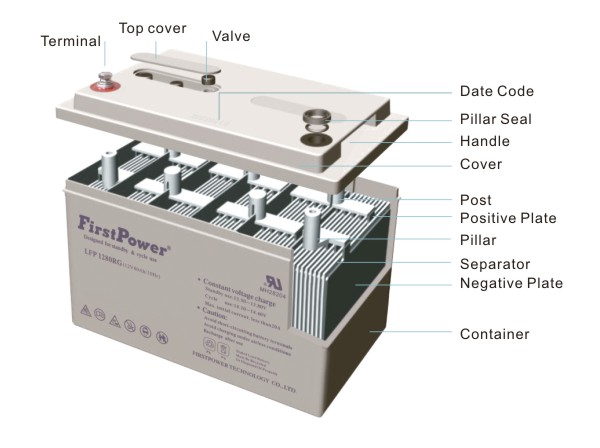
Lead-acid batteries are composed of a Lead-dioxide cathode, a sponge metallic Lead anode and a Sulphuric acid solution electrolyte. This heavy metal element makes them toxic and improper disposal can be hazardous to the environment.
The cell voltage is 2 Volts
As the work of lead acid battery is kind of complex, below we get a video to show the working process of it: